No-Stress Guide to pH and TDS
 In addition to proper lighting and ventilation, monitoring and controlling the pH and TDS of the nutrient solution is one of the most important aspects of a successful and productive grow room. Of the 100-plus elements on earth, a plant only needs less than 20 to survive. But a plant’s roots can only absorb those essential elements within a certain pH range (5.5 to 6.5 for hydroponics, 6.0 to 7.0 for soil). Additionally, even if those elements are in a nutrient solution at the correct pH, the roots will not be able to absorb them if they are at too high of a strength. If either measurement is off, nutrients may not be available to the plant when it needs them or they may be at such a high concentration that the nutrient solution actually reverses the flow of nutrients into the roots, causing damage to the plant. With that in mind, it is easy to see why measuring pH and TDS is so important for healthy plants, heavy harvests and happy growers
In addition to proper lighting and ventilation, monitoring and controlling the pH and TDS of the nutrient solution is one of the most important aspects of a successful and productive grow room. Of the 100-plus elements on earth, a plant only needs less than 20 to survive. But a plant’s roots can only absorb those essential elements within a certain pH range (5.5 to 6.5 for hydroponics, 6.0 to 7.0 for soil). Additionally, even if those elements are in a nutrient solution at the correct pH, the roots will not be able to absorb them if they are at too high of a strength. If either measurement is off, nutrients may not be available to the plant when it needs them or they may be at such a high concentration that the nutrient solution actually reverses the flow of nutrients into the roots, causing damage to the plant. With that in mind, it is easy to see why measuring pH and TDS is so important for healthy plants, heavy harvests and happy growers
What is pH and TDS?
pH is an abbreviation of for "potential hydrogen." It is a measurement of a solution's acidity or alkalinity that is expressed on a scale of 0 to 14, 0 being the most acidic, 14 being the most alkaline and 7 being neutral. The scale is based on logarithms, so moving up or down one full point represents a 10x increase or decrease in acidity or alkalinity (a solution at pH 5 is 10 times more acidic than a solution at pH 6 and 100 times more acidic than pH 7, for example).
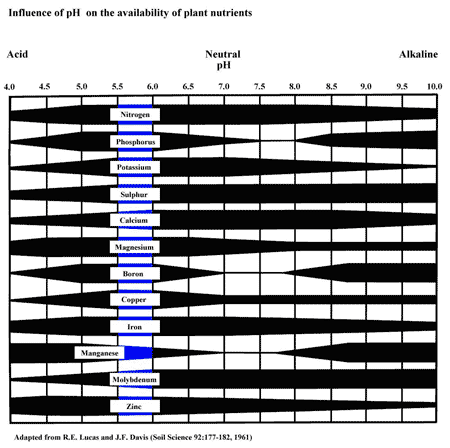
In hydroponics, the majority of elements a plant needs are available within the pH range of 5.5 and 6.0, as seen in the chart below.
TDS (Total Dissolved Solids) is a measurement of the strength of a nutrient solution. It can be expressed in PPM (Parts Per Million) or EC (Electrical Conductivity), which states values in µS (MicroSiemens) or mS (MilliSiemens). PPM tends to be the most common in the United States while EC is more common internationally.
PPM and EC meters measure nutrient strength in the same way with the same accuracy, but the readouts are displayed differently. For example, a nutrient solution that measures 1000 PPM on a TDS meter with a 0.7 conversion factor is the same strength as a solution that registers 1430 µS or 1.43 mS on an EC meter. PPM and EC meters measure the strength of a nutrient solution by measuring the flow of electrical current between two metallic probes. The higher the salt concentration in the nutrient solution, the better it conducts electricity.
Knowing your meter's conversion factor is also important when it comes time to calibrate it. Hanna calibration solutions are generally for meters with a 0.5 conversion factor while Genesis calibration solution is generally for meters with a 0.7 conversion factor.
Putting it to the test
TDS and pH of the nutrient solution is very important in hydroponics. Luckily, there are a variety of meters that read pH, PPM and EC.

One cost-effective way to check the pH of your nutrient solution is the Checker by Hanna Instruments. It is simple to use, easy to calibrate and has automatic temperature compensation.
Growers serious about getting their pH and PPM in check often turn to the Hanna Waterproof Combo Pen HI98129. With a rugged case and waterproof construction, the Combo Pen is a triple threat measuring pH, EC and PPM while automatically compensating for temperature. It also features automatic calibration and allows the user to change the conversion factor used for calculating PPM readings. For growing, the 0.7 conversion factor works well.
The water-resistant, handheld Hanna pH/EC/TDS/°C Portable Meter 9813-6N is a good choice for growers with larger operations who need fast and accurate readings of their nutrient solution. It features Hanna’s exclusive Cal Check function that allows the user to check the calibration of the meter at any time and calibration adjustments can be easily made using two knobs on the face of the meter. The 9813-6N also automatically compensates for temperature in pH, PPM and EC readings, and comes with a carrying case and sachets of pH and conductivity calibrations solutions.

For more demanding commercial operations that require the most rugged and convenient of meters, the Hanna Commercial ph/EC/TDS/Temp Meter HI991300N is the best bet. This commercial-grade meter is completely shielded against electrical interference, which means it gives precise readings even in the presence of ballasts, water pumps and other electronic equipment that can throw off readings. It lets the user select from a variety of calibration buffers to better adapt the meter to the job at hand, and its TDS calibration factor can be adjusted from 0.45 to 1.0.
Adjusting pH
If the pH of your nutrient solution is out of whack, you’ll need to get it back in range using either pH Down or pH Up. A small amount of these products can significantly raise or lower pH levels. To avoid constantly drifting past the desired pH level when adjusting pH, it's best to add very small amounts of pH Up or pH Down to gradually increase or decrease the pH level rather than adding too much pH Down and having to add pH Up to bring the solution back within range, or vice versa.
Growers using tap water may find it more difficult to adjust pH levels due to the buffering capability of calcium, magnesium and many other compounds commonly found in municipal water sources. A tap water-based nutrient solution will likely require a higher dose of pH down than a solution using RO water, since tap water tends to be on the alkaline side with a pH of 7.5 to 8.5 or higher, depending on the location.
Tap water will also make it more difficult to follow manufacturers' nutrient schedules, most of which set PPM levels based on zero-PPM source water. Growers seeking precise measurements should consider a reverse osmosis water filter.

pH and Synthetic Nutrients
Synthetic hydroponic nutrients, such as Botanicare's CNS 17, Canna's Aqua and Coco lines, or House & Garden's Aqua Flakes and Cocos nutrients, deliver nutrition in ionic form, meaning it is readily available to the plant and does not need to be broken down by microorganisms before roots can absorb it.
Synthetic nutrients can trigger a quicker response when fed to plants, and since those elements are available right away it becomes important that the nutrient solution is delivered to the plant at the proper pH to avoid nutrient lockout or having too much of one element available at the expense of other elements.
Adjusting pH with Natural and Organic Nutrients
When growing in soil with reverse osmosis water, adjusting pH is not necessary with some nutrient lines, such as General Organics or Canna Bio Vega, Bio Flores and Bio Boost. The natural ingredients in General Organics nutrients put beneficial bacteria in the root zone in charge of regulating pH and making nutrients available to the plant, while Canna’s Bio line contains compounds that makes its ingredients absorbable over a broad pH range.
However, not all organic nutrients are the same. The makers of BioBizz recommend adjusting the pH of the water to 7.0 before adding their products.
- The makers of BioBizz recommend adjusting the pH of the water to 7.0 before adding their products.
- For growers mixing Earth Juice nutrients into a reservoir, the manufacturer recommends taking a pH reading right after mixing the nutrients and taking another reading 24 hours later before adjusting the pH incrementally if the nutrient solution is not within range.
- Humboldt Nutrients suggests a pH range between 5.5 and 7.0 will work with their Bloom and Grow Natural line. These nutrients contain a number of chelating agents that allow the elements to be absorbed by the plant within a broader range of pH values.
Since PPM and EC readings are based on the amount of mineral salts in a nutrient solution, PPM or EC readings will not be very telling when measuring a solution made up of organic nutrients, which have very little if any mineral salts. To ensure proper nutrient strength with organic nutrients, follow the manufacturer’s recommended feeding schedule, and watch plants for signs of deficiency or over-fertilization.
